Diamagnetic materials move away from a magnet. Paramagnetic materials move toward a magnet.
This is the quick answer people want when they search “diamagnetic or paramagnetic.”
Magnets affect many materials. Some objects move a little away from a magnet. Some move a little closer. The movement is very small. But scientists can measure it.
The reason comes from electrons. Electrons live inside atoms. They move and spin. This spin makes a tiny magnetic force.
In some materials, electrons stay in pairs. The pair cancels the magnetic force. The material then moves away from a magnet. This is called diamagnetic behavior.
In other materials, some electrons stay alone. These are called unpaired electrons. They create a small magnetic force. The material moves toward the magnet. This is called paramagnetic behavior.
Students learn these ideas in physics and chemistry. Scientists also use them to study matter and build new materials.
In this guide, you will learn the meaning of diamagnetic or paramagnetic, their origin, key differences, and real examples.
📖 Diamagnetic vs Paramagnetic Definition
🔵 Diamagnetic Definition
Diamagnetic materials move slightly away from a magnet.
All electrons in these materials are paired.
Each pair cancels the magnetic force.
Because of this, the material shows weak repulsion.
Diamagnetic materials do not become permanent magnets.
Examples of Diamagnetic Materials
| Material | Common Use |
| Copper | Electrical wires |
| Gold | Jewelry |
| Silver | Electronics |
| Water | Science experiments |
| Bismuth | Magnetic tests |
Example sentence
Copper shows diamagnetic behavior in a strong magnetic field.
🔴 Paramagnetic Definition
Paramagnetic materials move slightly toward a magnet.
These materials have unpaired electrons.
Unpaired electrons create small magnetic forces.
When a magnet appears, these electrons follow the magnetic field.
This creates weak attraction.
Paramagnetic materials lose this magnetism when the magnet is removed.
Examples of Paramagnetic Materials
| Material | Common Use |
| Aluminum | Aircraft parts |
| Magnesium | Manufacturing |
| Oxygen | Medical use |
| Titanium | Aerospace |
| Platinum | Chemical catalysts |
Example sentence
Aluminum shows paramagnetic behavior near a magnet.
⚡ Diamagnetic and Paramagnetic : Quick Answer
The difference depends on electrons and magnetic reaction.
| Feature | Diamagnetic | Paramagnetic |
| Electron type | All paired | Some unpaired |
| Magnet reaction | Moves away | Moves closer |
| Magnetic strength | Very weak | Weak |
| Permanent magnet | No | No |
Simple idea:
- Diamagnetic → Moves away from magnet
- Paramagnetic → Moves toward magnet
🕰️ Origin of Paramagnetic and Diamagnetic
Diamagnetic Origin
The word diamagnetic comes from Greek.
Dia means across.
Magnetism means magnetic force.
Scientists used this term for materials that push away magnetic fields.
The effect was studied in the 1800s.
Michael Faraday helped explain it.
Paramagnetic Origin
The word paramagnetic also comes from Greek.
Para means beside.
Scientists used this word for materials that respond to magnets.
Later studies showed that unpaired electrons cause this effect.
🌍 Key Differences Between Diamagnetic and Paramagnetic
Diamagnetic and paramagnetic materials behave in opposite ways.
| Property | Diamagnetic | Paramagnetic |
| Magnet effect | Repelled | Attracted |
| Electron pairing | All paired | Some unpaired |
| Magnetic sign | Negative | Positive |
| Magnetic strength | Very weak | Weak |
Both effects are small.
Scientists often use strong magnets to observe them.
🔬 Examples of Diamagnetic and Paramagnetic Materials
Diamagnetic Examples
Diamagnetic materials move away from magnets.
| Material | Field |
| Copper | Electronics |
| Silver | Conductors |
| Gold | Jewelry |
| Water | Chemistry |
| Graphite | Industry |
These materials show weak magnetic repulsion.
Paramagnetic Examples
Paramagnetic materials move toward magnets.
| Material | Field |
| Aluminum | Engineering |
| Magnesium | Manufacturing |
| Oxygen | Chemistry |
| Titanium | Aerospace |
| Platinum | Catalysts |
These materials show weak magnetic attraction.
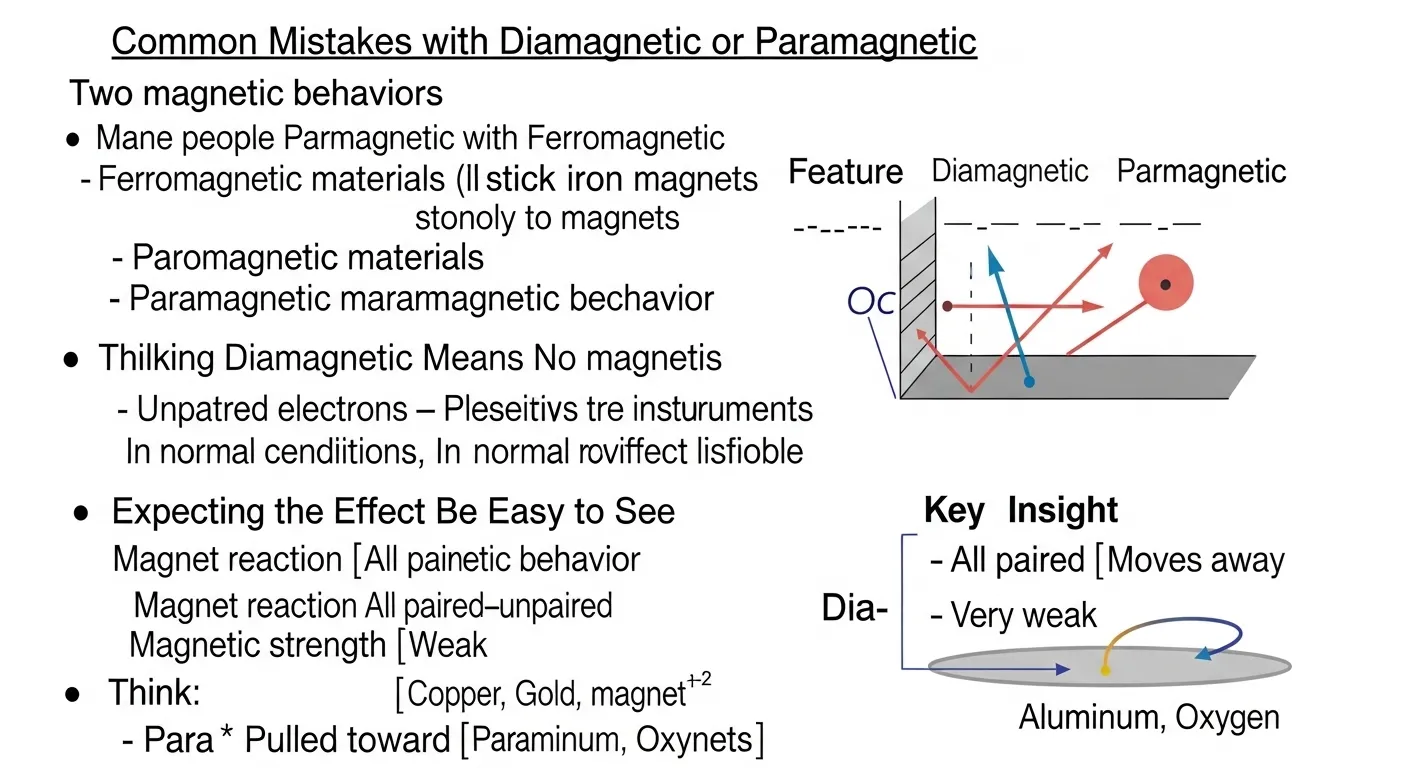
⚠️ Common Mistakes with Diamagnetic or Paramagnetic

Confusing paramagnetic with ferromagnetic
Iron is ferromagnetic.
It sticks strongly to magnets.
Paramagnetic materials do not.
Their attraction is weak.
Thinking diamagnetic means no magnetism
Diamagnetic materials still react to magnets.
The reaction is simply very small.
Ignoring electron pairing
Electron pairing explains magnetic behavior.
Expecting large movement
The movement is very small.
Scientists often use strong magnets to measure it.
💬 Diamagnetic or Paramagnetic in Everyday Examples
Classroom learning
Students often ask if aluminum is diamagnetic or paramagnetic.
Science research
Researchers study paramagnetic materials in medical imaging.
Educational content
Teachers explain diamagnetic and paramagnetic behavior in chemistry classes.
Laboratory work
Scientists place materials in magnetic fields to test their properties.
📊 Diamagnetic or Paramagnetic: Keyword Variations
| Keyword | Meaning |
| diamagnetic vs paramagnetic | Comparison search |
| paramagnetic definition | Concept explanation |
| diamagnetic examples | Learning topic |
| magnetic properties of elements | Chemistry concept |
These keywords are common in student searches.
❓ FAQs About Diamagnetic or Paramagnetic
What is the main difference between diamagnetic and paramagnetic?
Diamagnetic materials move away from magnets. Paramagnetic materials move toward magnets.
Why are diamagnetic materials repelled?
All electrons are paired. Their magnetic forces cancel.
Why are paramagnetic materials attracted?
They contain unpaired electrons that follow the magnetic field.
Are paramagnetic materials strong magnets?
No. Their attraction is weak.
Is water diamagnetic?
Yes. Water is a diamagnetic substance.
Is oxygen paramagnetic?
Yes. Oxygen has unpaired electrons.
Can temperature affect paramagnetic materials?
Yes. High temperature can reduce magnetic alignment.
Conclusion
Diamagnetic and paramagnetic materials show different magnetic behavior. The difference comes from electrons.
Diamagnetic materials contain only paired electrons. Their magnetic forces cancel. Because of this, the material moves slightly away from a magnet. Copper, gold, and water are examples.
Paramagnetic materials contain unpaired electrons. These electrons create small magnetic forces. When a magnetic field appears, the electrons align with the field.
This causes the material to move slightly toward the magnet. Aluminum, oxygen, and magnesium are examples.
Both types show weak magnetic effects. They are not strong magnets like iron. Still, these properties are important in science. Scientists use them to understand atoms, electrons, and magnetic forces.
Learning the difference between diamagnetic or paramagnetic materials helps explain how magnetism works inside matter.
Discover More Articals!
🌏 Vietnam or Thailand: Which Destination Is Better?
🌾 Artisan or Agriculturist: Meaning, Differences and Explained
Velo or Zyn : Which Nicotine Pouch Should You Choose?